Electrochemistry
Electrochemical Cells: Concentration Cells Demonstration and Simulation
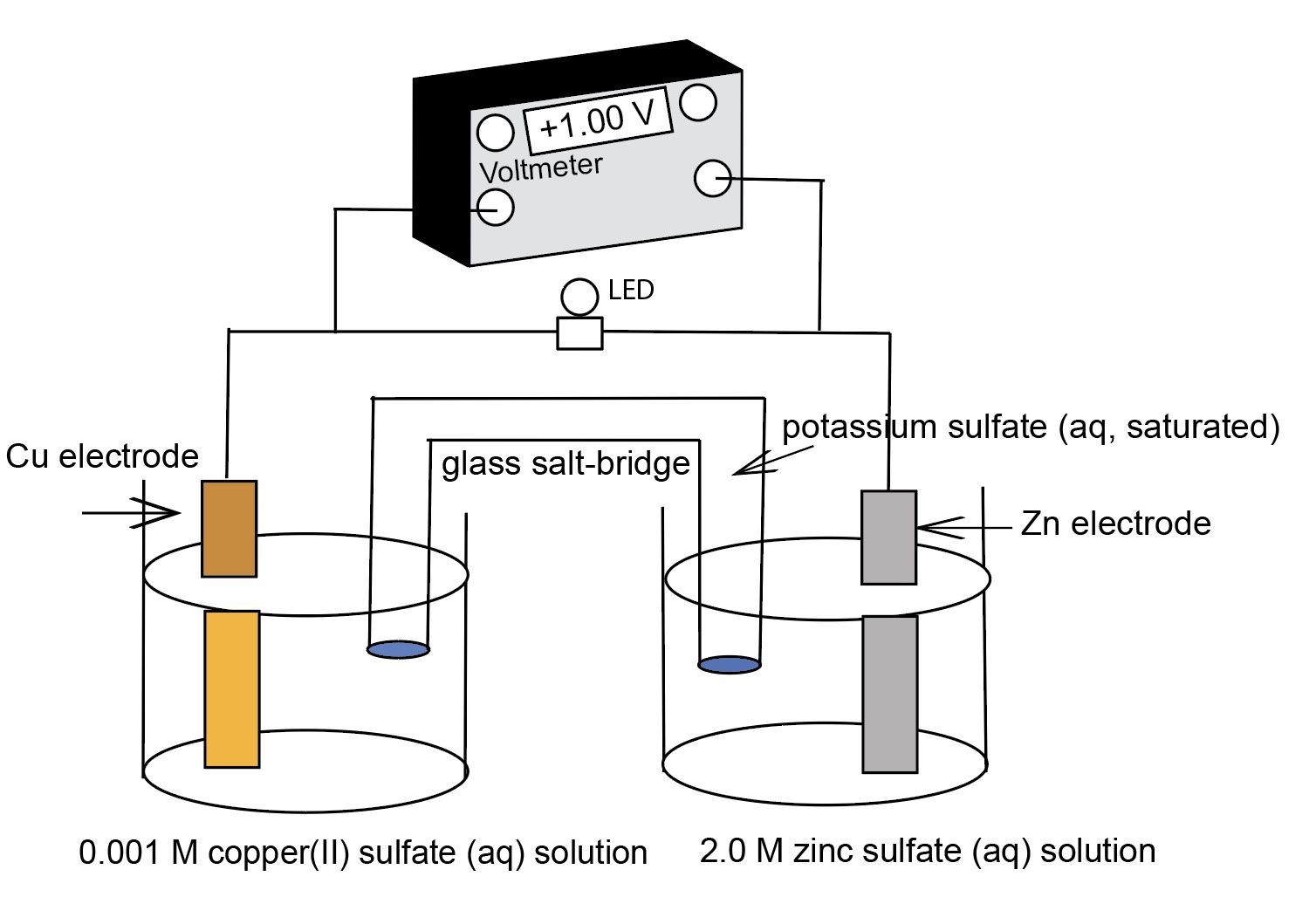
This presentation illustrates how chemical reactions can generate electricity. Electrochemical cells based on an electromotive force (emf) generated by virtue of a difference
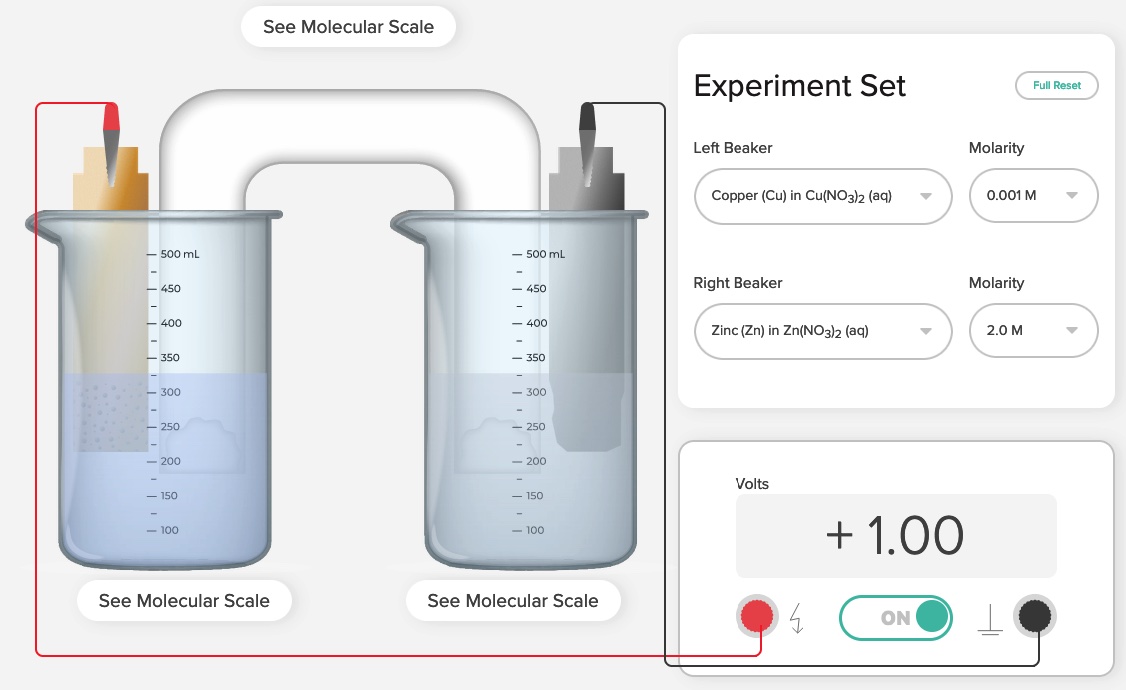
Nernst Electrochemical Cells Simulation AACT
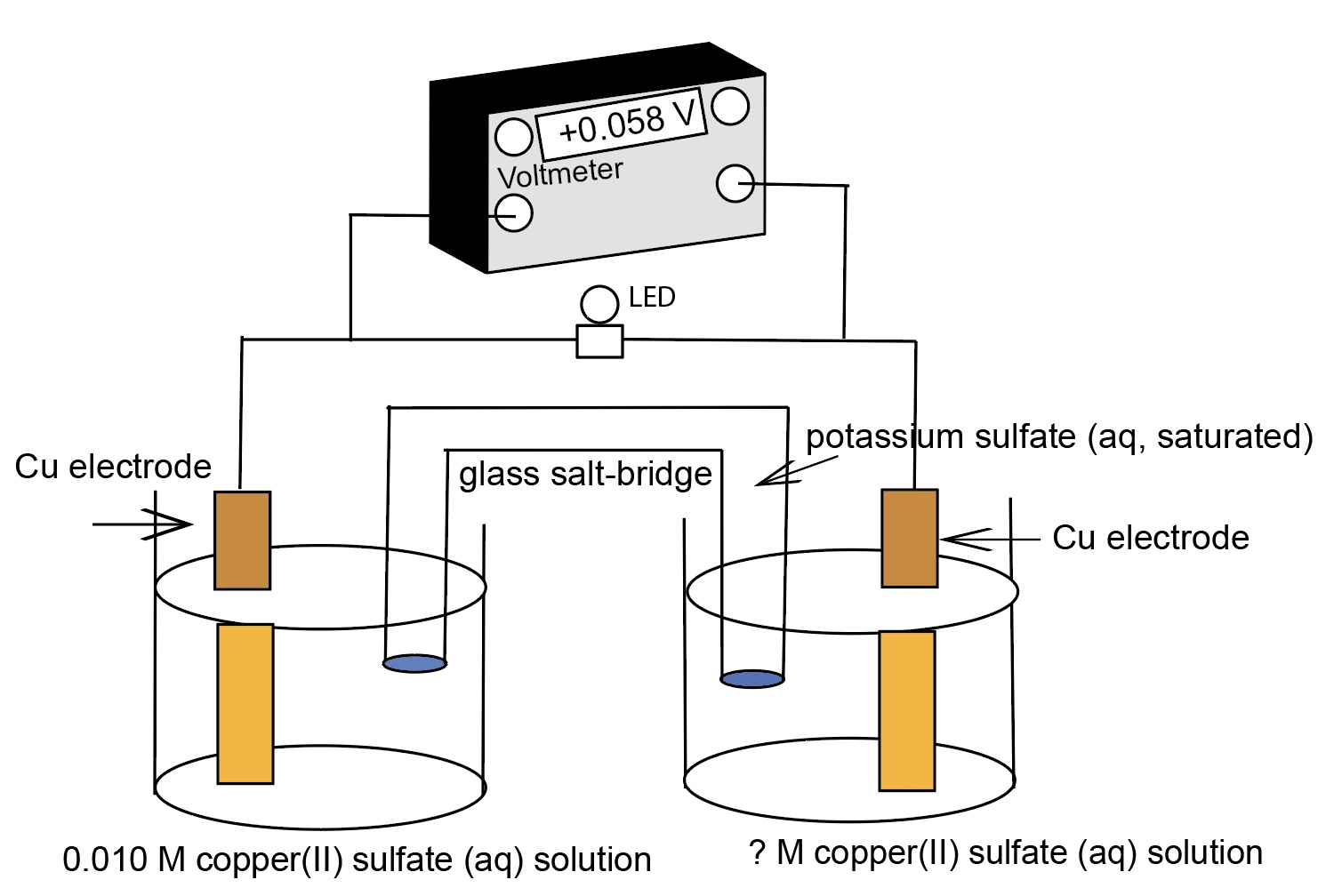
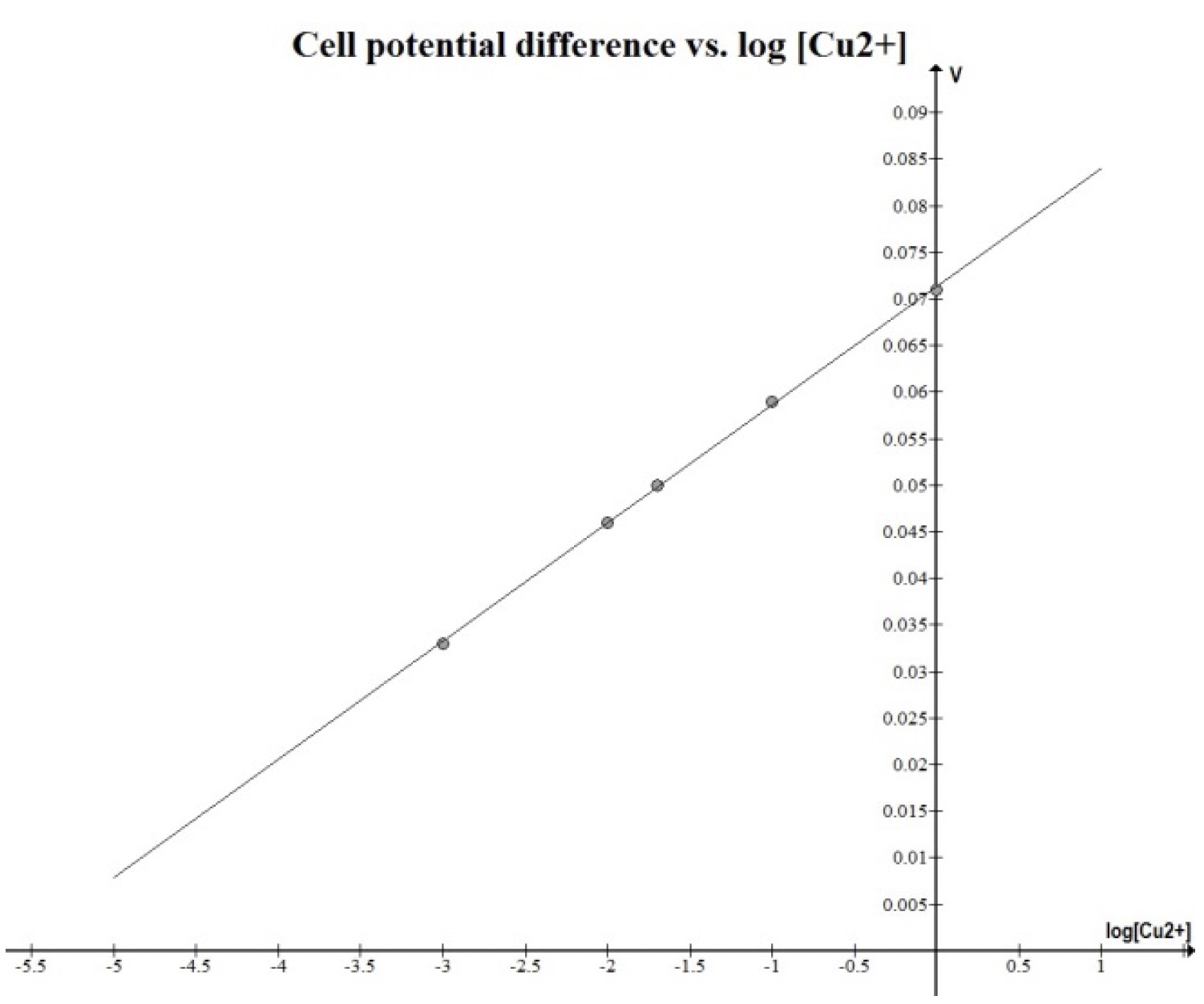
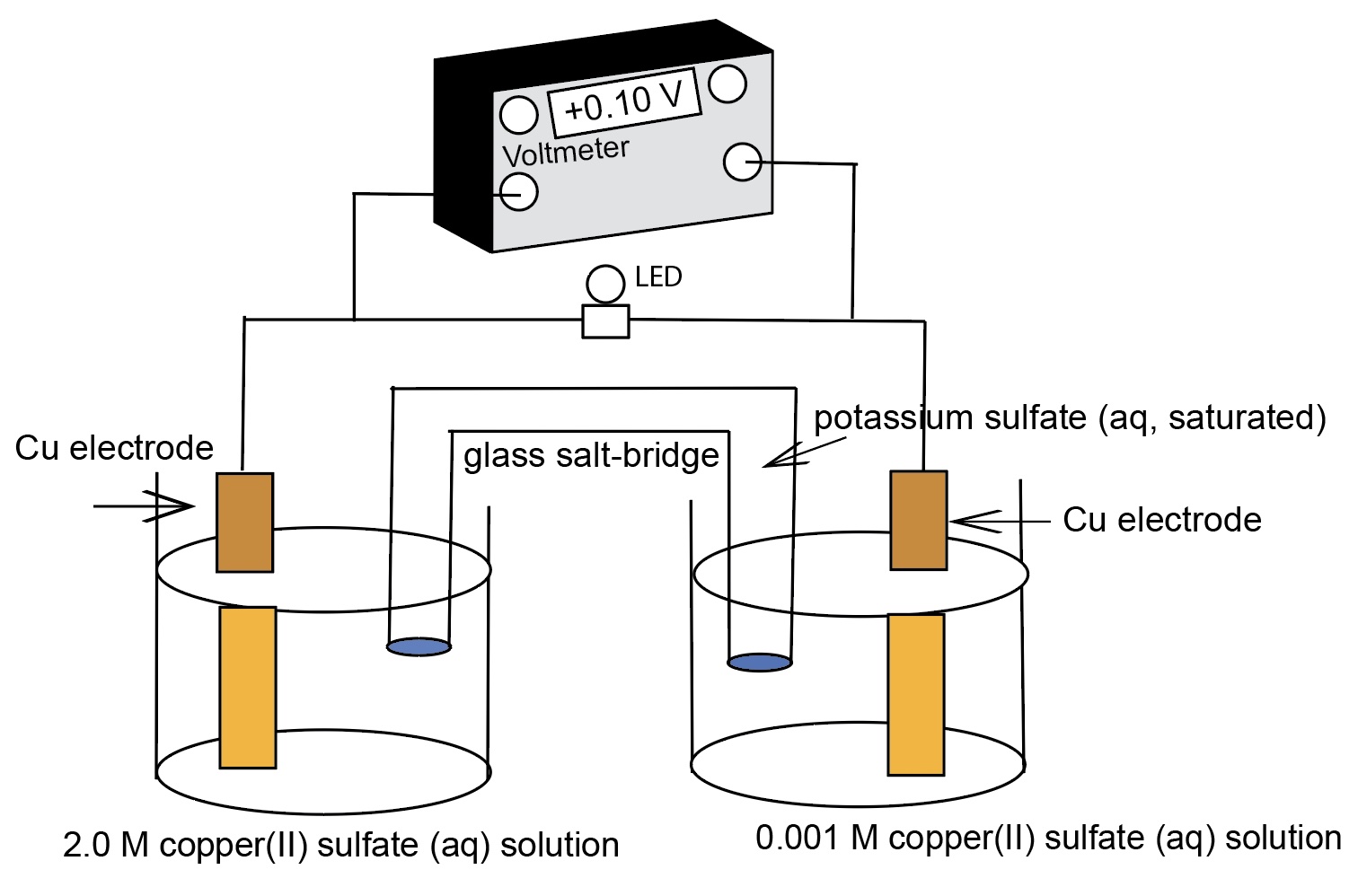
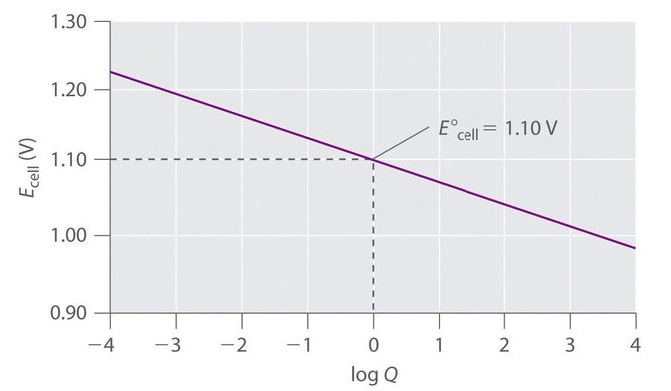
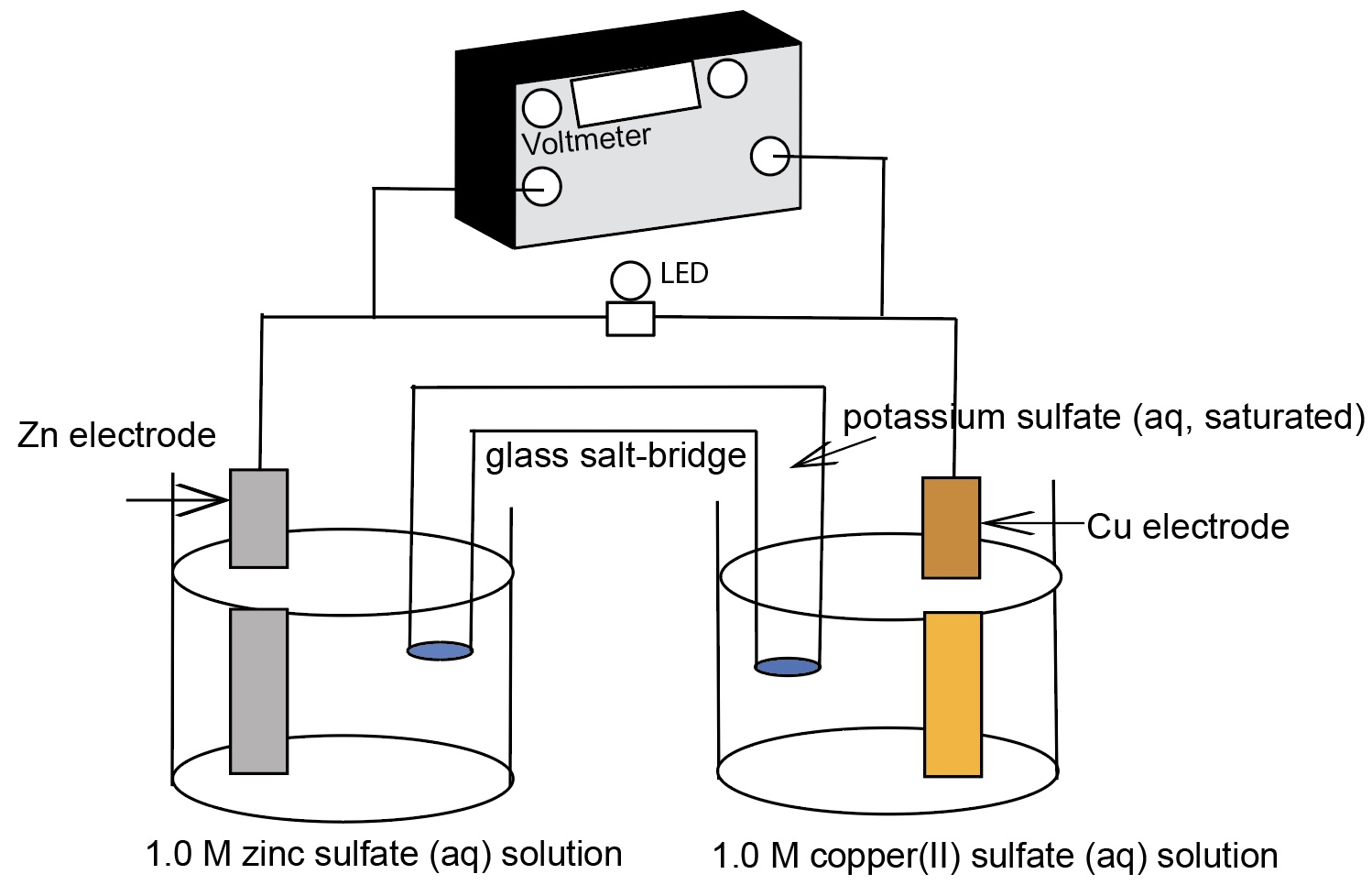
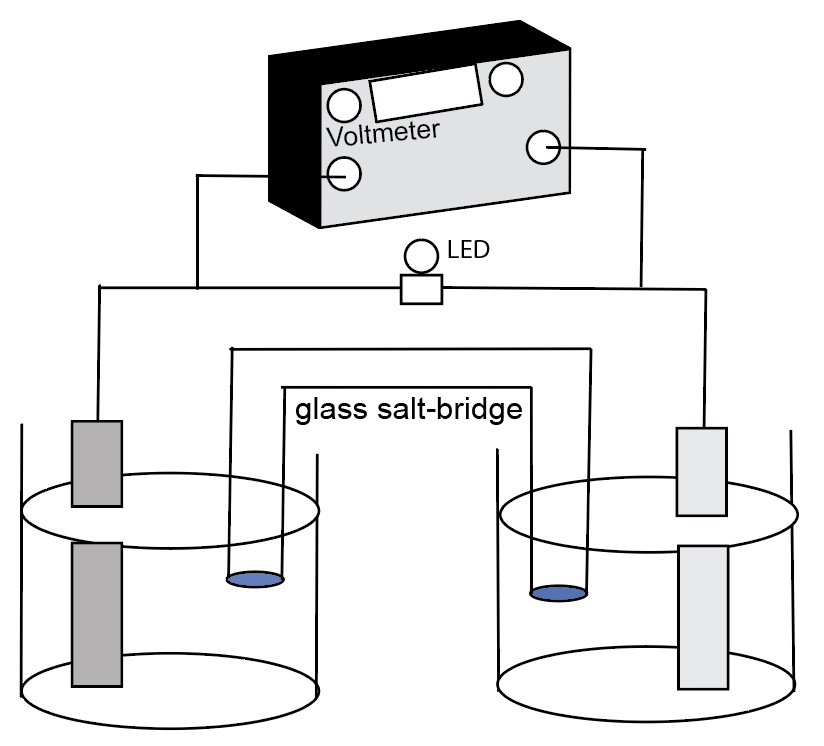
Nernst Cells. The dependence of the cell potential on the concentration of the ions in each of the half-cells is linked directly to the dependence of free energy,
Electrochemical Cells Demonstration: Non-Standard Cells Nernst Equation AACT Simulation
This presentation illustrates how chemical reactions can generate electricity. This presentation uses a series of real concentration electrochemical cell and a series of conce
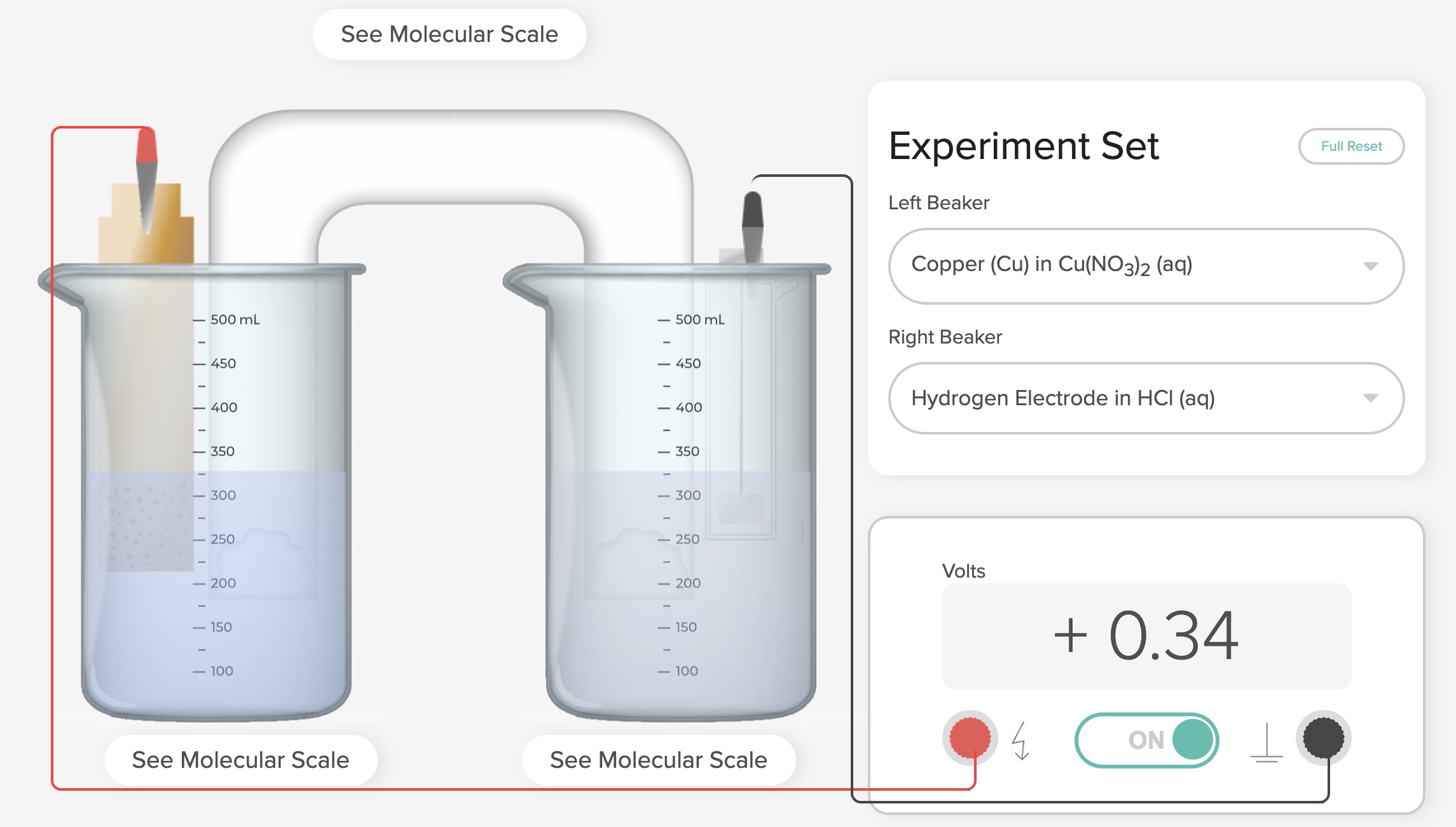
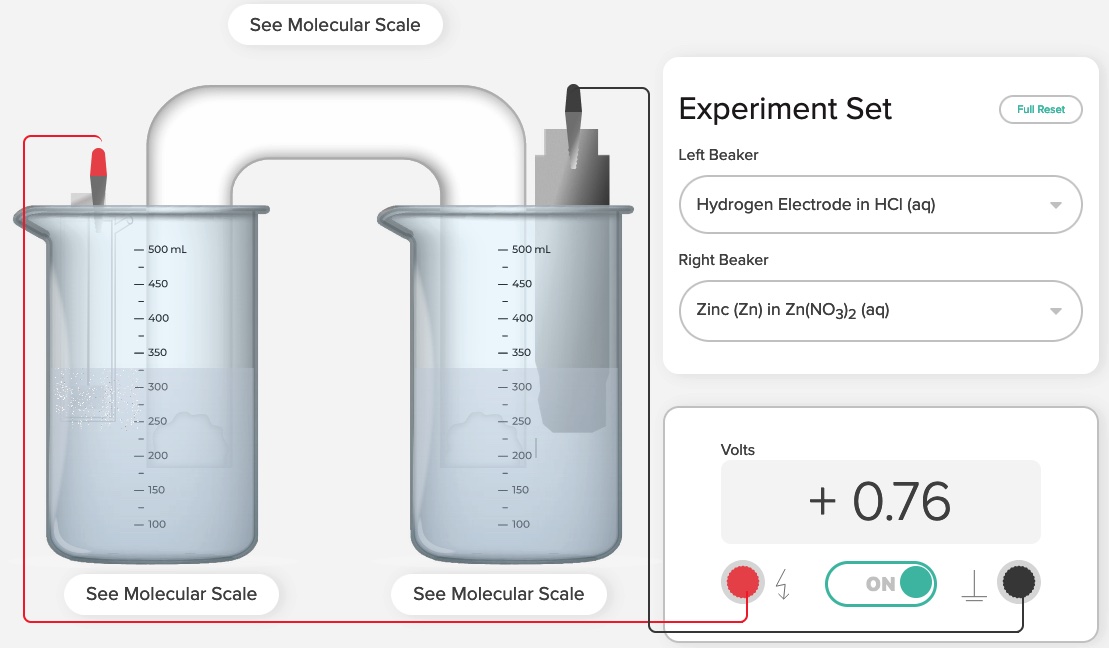
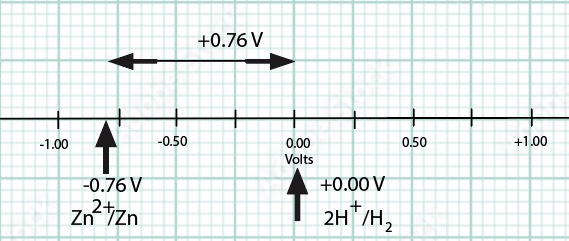
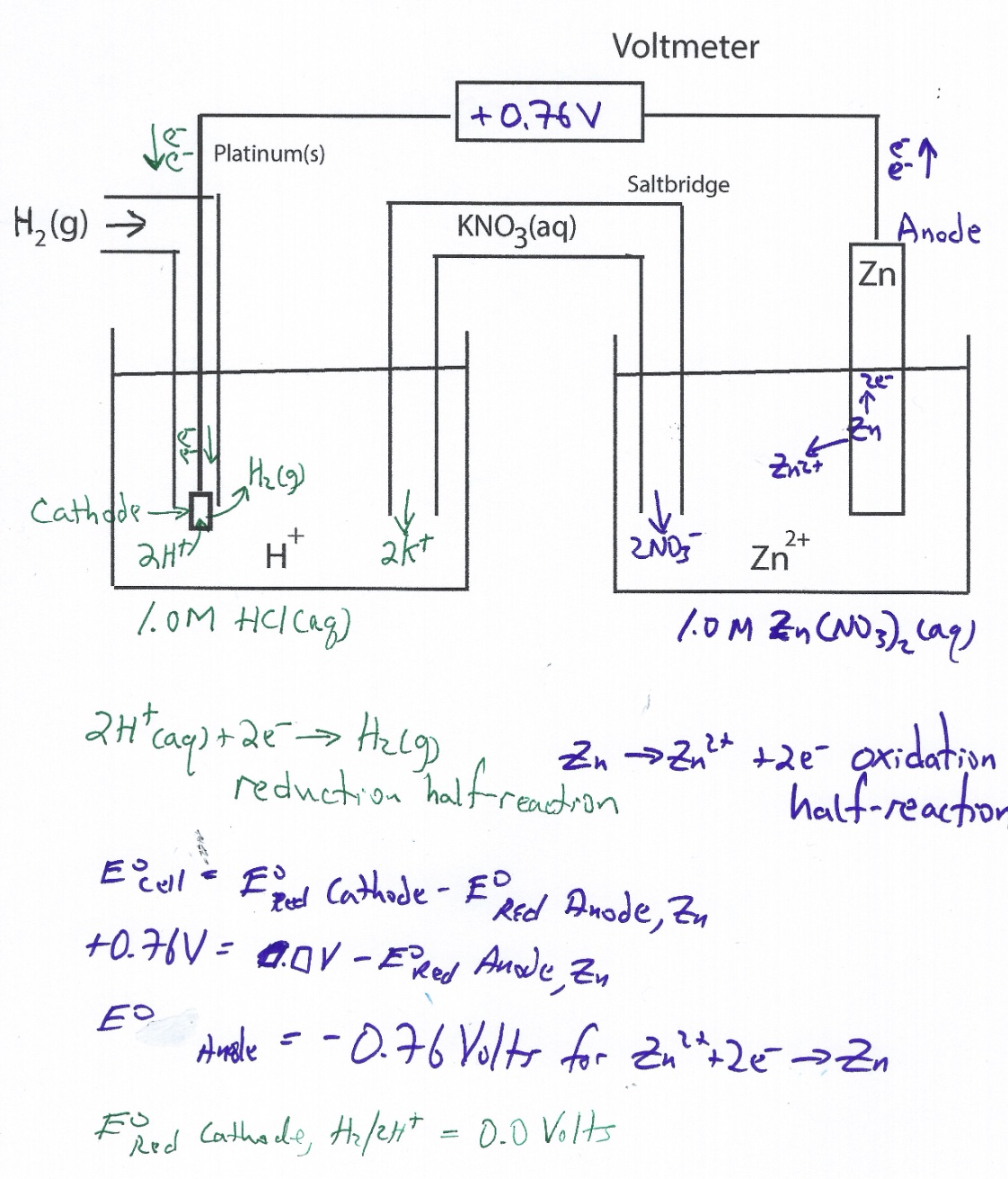
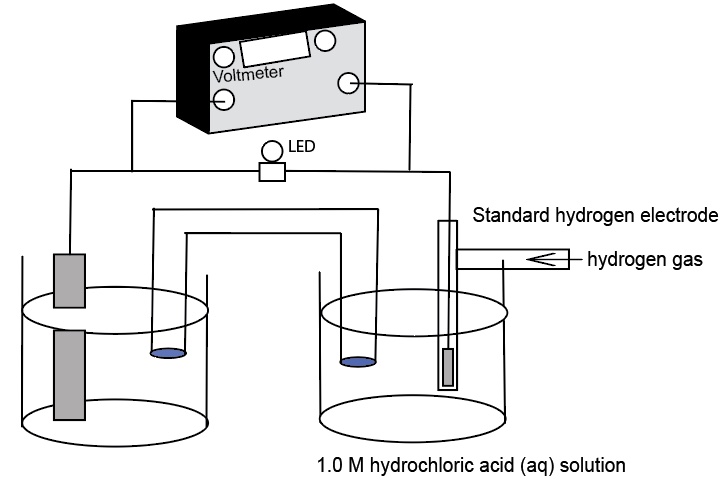
Standard Hydrogen Electrode Demonstration and AACT Simulation
A Standard Hydrogen Electrode (SHE) is used to compare the force of the flow of electrons i
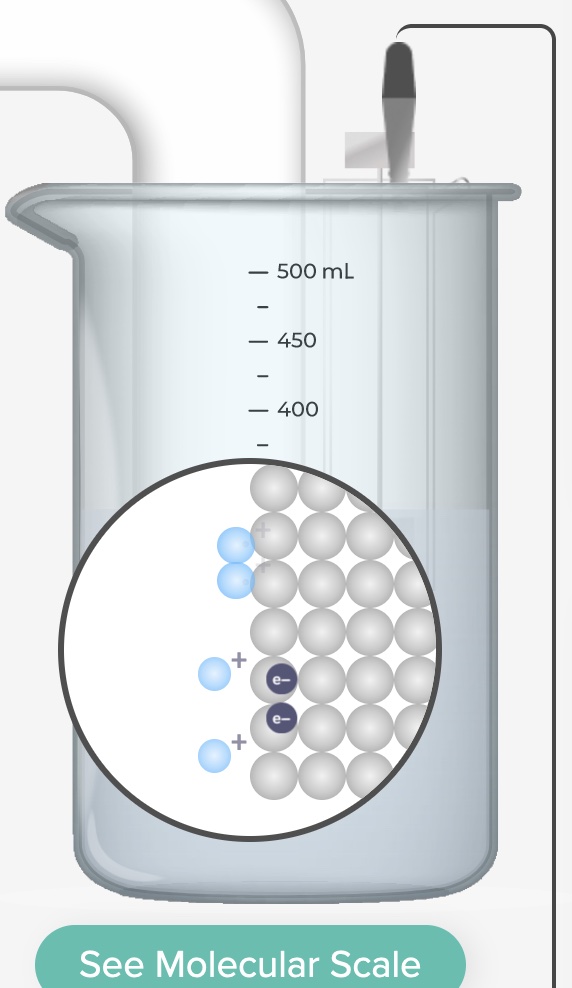
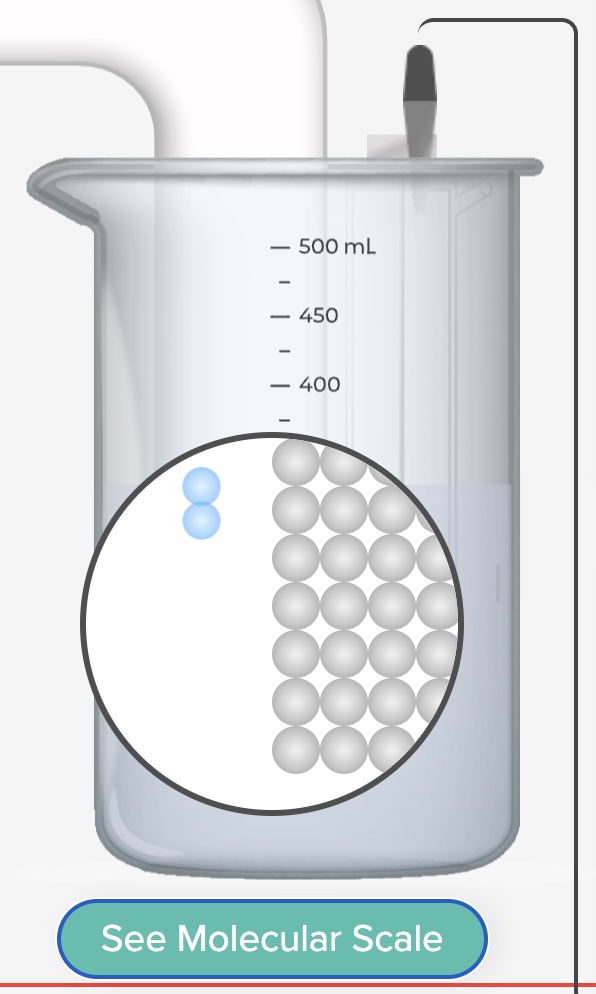
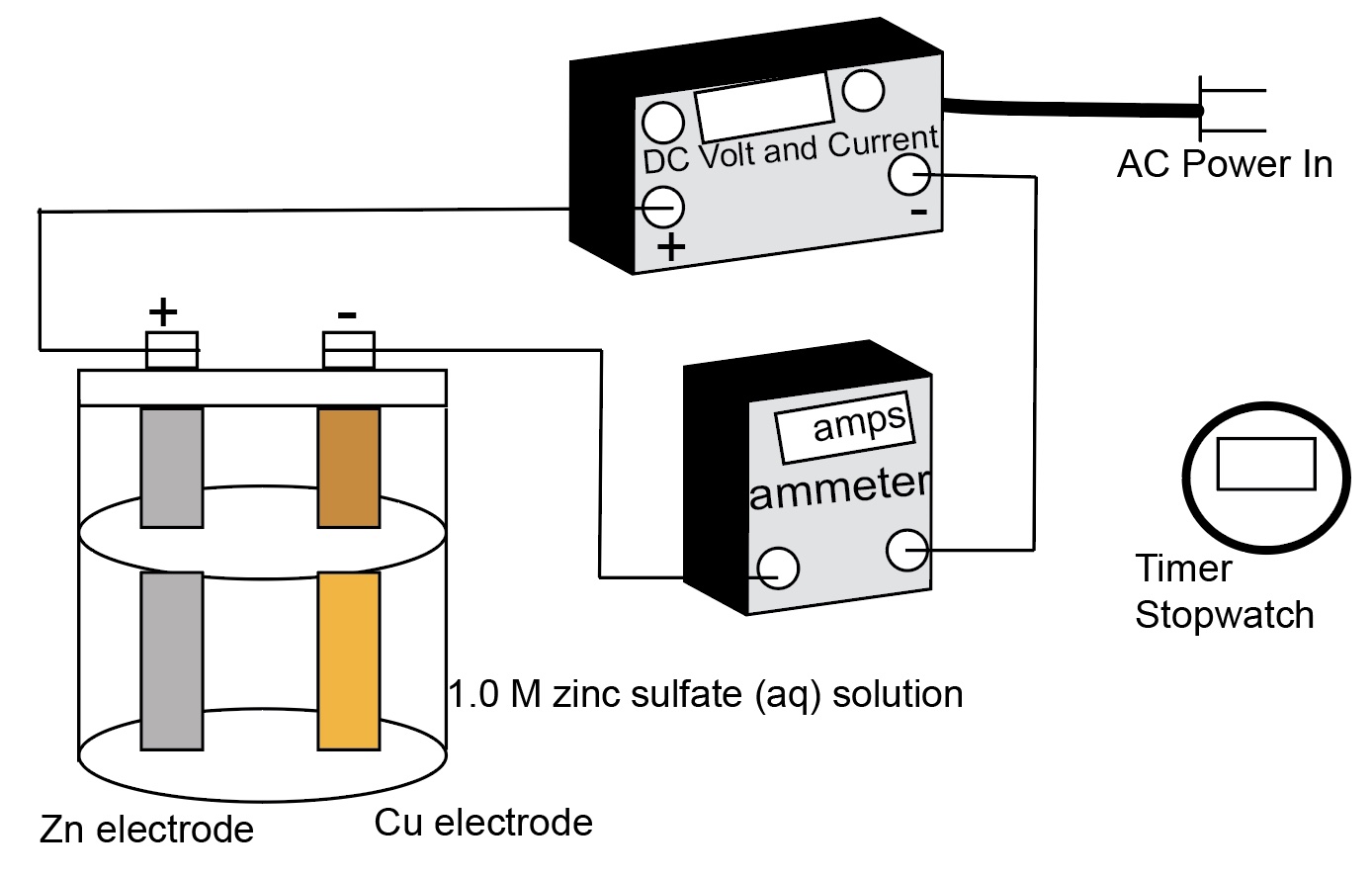
Electrolysis: Plating Zinc Metal on Copper Real and Simulation
Plate zinc metal on copper using electrolysis. Demonstration of a real electrolysis cell accompanied by a computer simulation of an electrolysis cell.
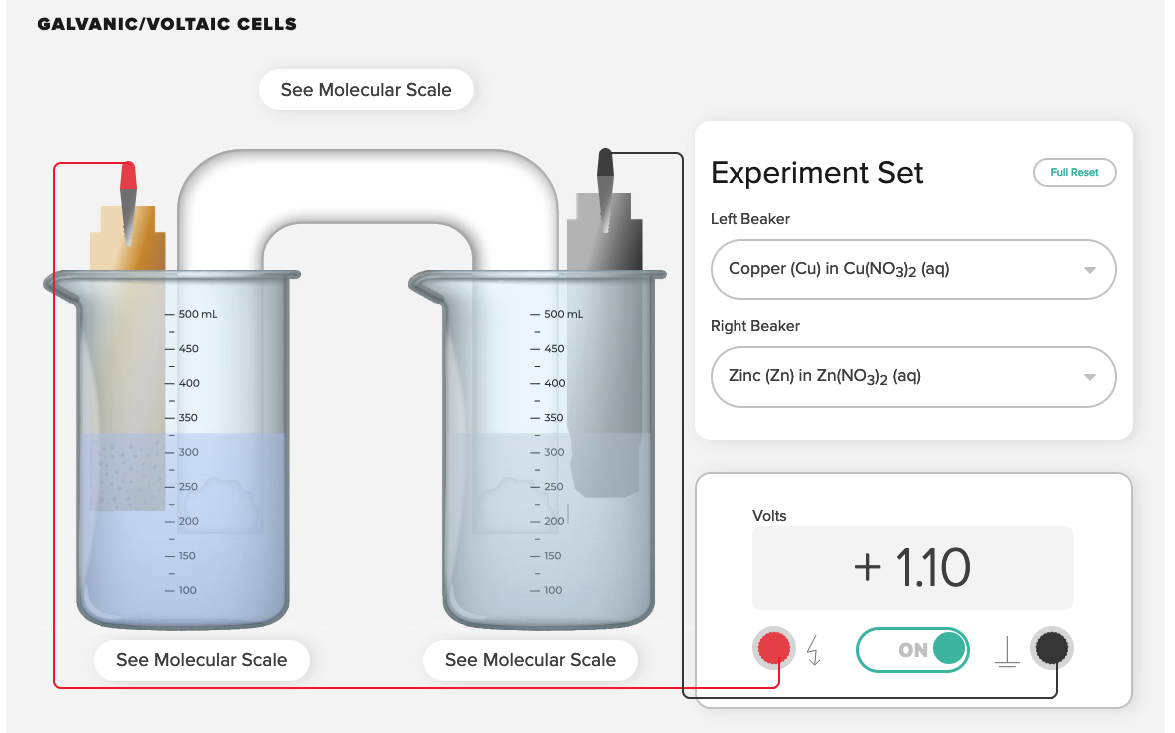
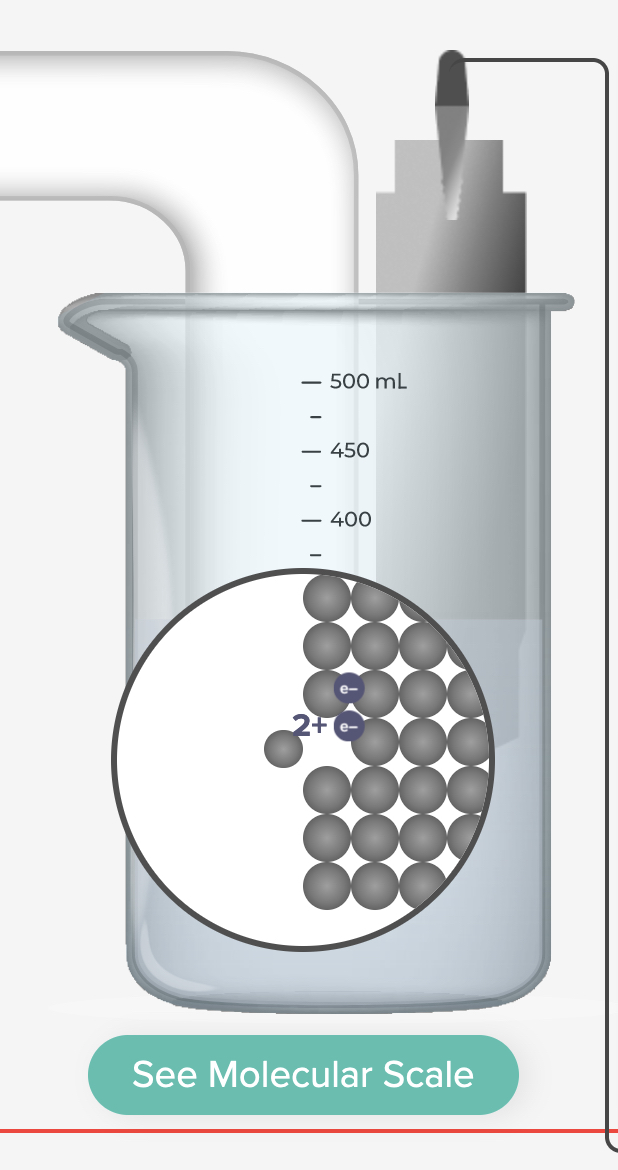
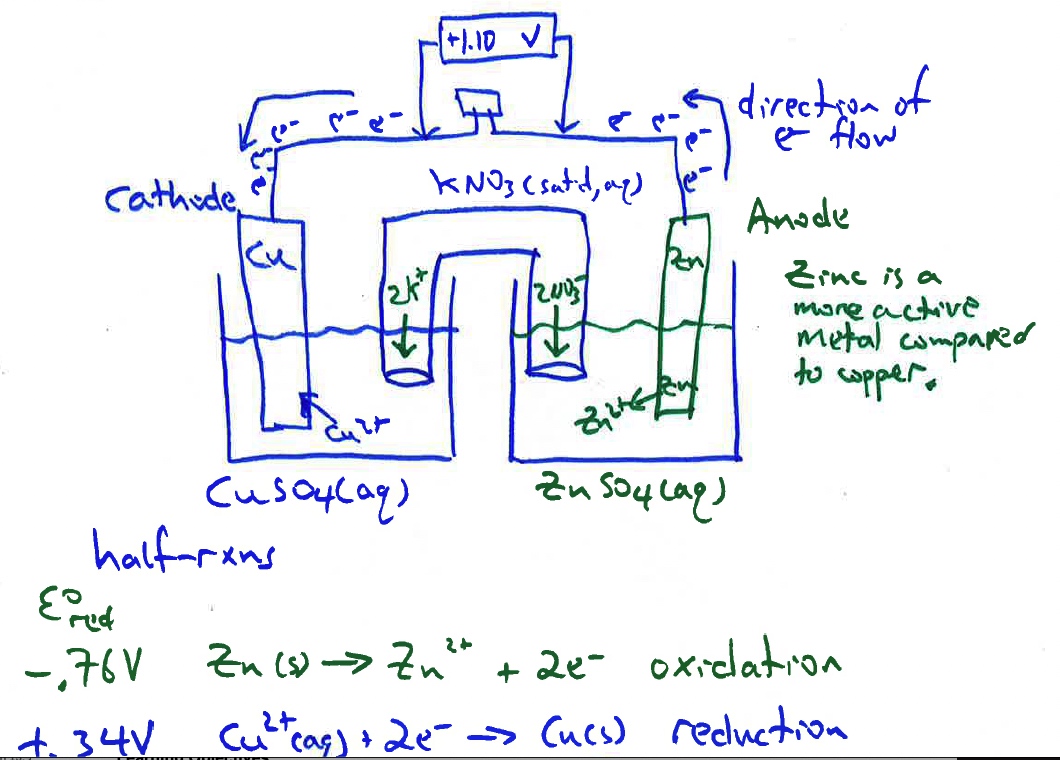
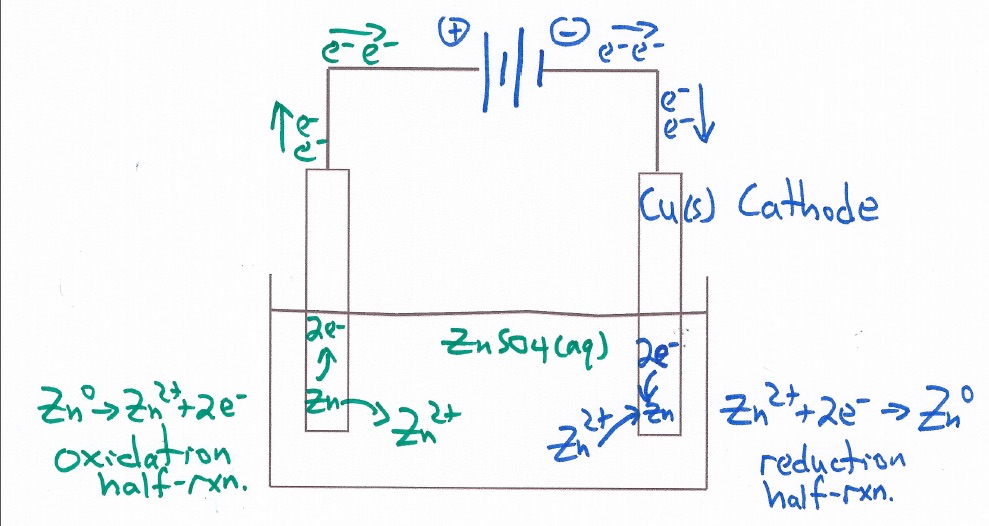
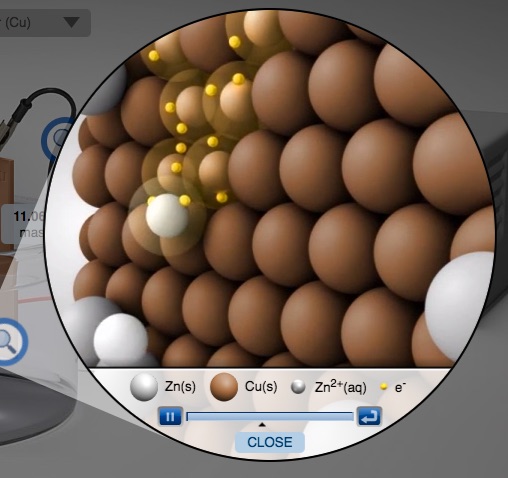
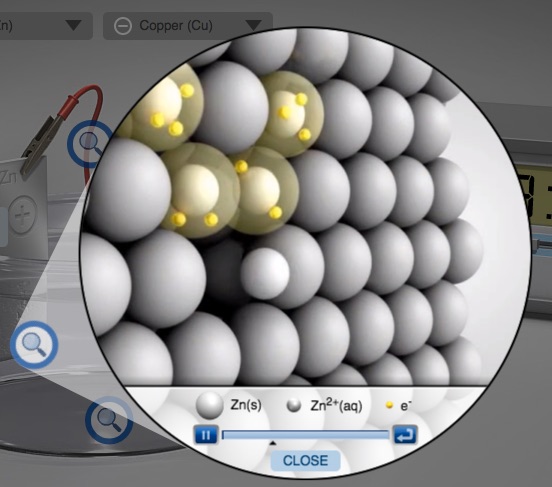
Electrochemical Cell Zinc Copper Demonstration and AACT Simulation Galvanic (Voltaic) Cells
In this interactive activity, the instructor demonstrates several aspects of galvanic cells using real electrochemical cells and a computer simulation.
Standard Hydrogen Electrode (SHE) Simulation and Animation AACT
The Voltaic Cell computer simulation is used to measure the E°cell generated by an electrochemical cell incorporating a standard hydro
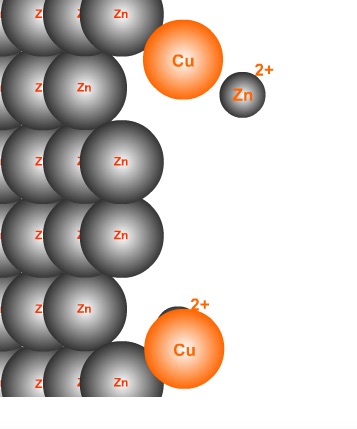
Single Displacement Reaction: Zinc and Copper(II) Ion REDOX
Co-Authors: T. Greenbowe and R. Sullivan This page is under construction.